|
The atomic mass (or the atomic weight) of an element may now be defined as the average relative mass (or weight) of its atom compared with one atom of carbon taken as 12.  
This unit called the atomic mass unit (a.m.u) is defined as 1/12 the mass of one 12C atom. Thus, the present unit for expressing atomic masses of the elements is based on the isotope of carbon ( 12C) which has six protons and six neutrons in its nucleus. This isotope of carbon has been assigned a value of exactly 12 atomic mass units (a.m.u) and is designated as 12C (or C 12) or carbon-12. To overcome any difficulty arising out of this duality, the International Union of Pure and Applied Chemistry and the International Union of Pure and Applied Physics decided, in 1961 to take lighter of the two naturally occurring isotopes of carbon as the standard of mass. Now as naturally occurring oxygen consists of three isotopes (mass numbers 16, 17, 18), the average value for the atomic mass of oxygen was taken as 16.0044 on the physical scale. The atomic mass of oxygen was fixed at exactly 16. For certain reasons (example- oxygen combines with the largest number of elements), oxygen was found to be much more convenient element as a standard than hydrogen. For example- oxygen atom was found 15.88 times that of the hydrogen atom. The amu is an older unit, and has been replaced by the unified atomic mass unit. The atomic mass unit is defined as 1/12 of the mass of the. atomic mass unit A unit in mass spectrometry that represents the relative scale, in which the mass of 1H is given an integral value of 1, 12C is 12, and so on. The same concept can be extended to ionic compounds and molecules. Thus, since the atomic mass of iron is 55.847 amu, one mole of iron atoms would weigh 55.847 grams. Atomic masses of other elements indicated how many times their one atom was heavier than the hydrogen atom. Atomic mass unit is a mass unit and is used to express the mass of atoms and other particles. The atomic mass is useful in chemistry when it is paired with the mole concept: the atomic mass of an element, measured in amu, is the same as the mass in grams of one mole of an element. To express the masses of atoms conveniently, hydrogen, the lightest of all elements, was selected as the standard and it was given the atomic mass equal to unity. It has, however, been found convenient to express the masses of atoms in terms of a special unit called the atomic mass unit. This indicates that gram is too big a unit to weigh an atom and hence to express the weight of an atom in gram is unreasonable. For instance, the masses of hydrogen, oxygen and silver atoms are 0.1673 X 10 -23 g, 2.657 X 10 -23 g and 17.9 X 10 -23 g, respectively. One atomic mass is also called one Dalton (Da) or unified. Most of the mass of an atom is concentrated in the protons and. It is so small and so light that it cannot be weighed by any laboratory balance. Atomic Mass Unit is defined as a mass exactly equal to one-twelve the mass of one carbon-12 atom. atomic mass, the mass of a single atom, usually expressed in atomic mass units (amu). 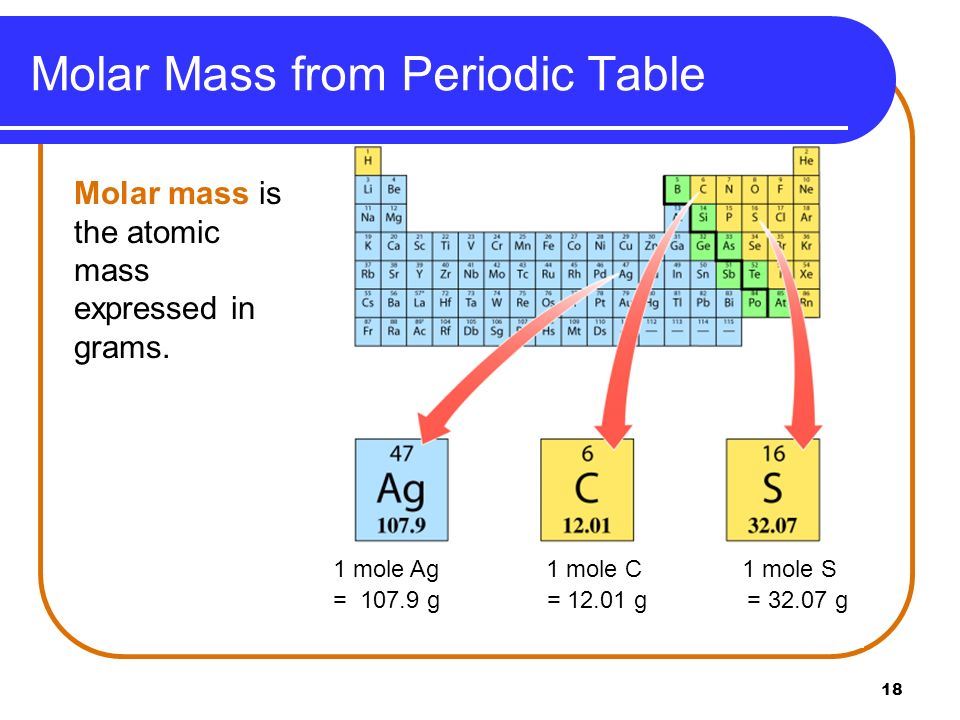
An atom is the smallest particle that can take part in a chemical reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
